CTC
What is CTC?
Cancer cells that detach from the primary tumor mass and enter the bloodstream are called circulating tumor cells (CTCs). While the majority of CTCs are destroyed by the immune system and some die on their own, a small number survive. Among these surviving CTCs, some travel through the bloodstream to reach a new organ or tissue, proliferate there, and cause metastasis, which is responsible for 90% of cancer-related deaths.
What is CTC Liquid Biopsy?
CTC liquid biopsy is a method that enables the capture and analysis of circulating tumor cells (CTCs) from the blood of cancer patients.
Purposes of performing CTC liquid biopsy include:
Cancer diagnosis
Planning personalized treatment
Monitoring treatment effectiveness
Detecting cancer recurrence
What Are the Advantages Compared to Tissue Biopsy?
In traditional tissue biopsy, which is commonly used in cancer diagnosis and monitoring, all or part of the tumor tissue is removed and analyzed.
The CTC liquid biopsy technique, on the other hand, only requires drawing a tube of blood, offering many advantages to both patients and physicians compared to surgical tissue biopsy.
Advantages for the patient:
Enables close monitoring of the disease and treatment response
Carries no risk
Causes no pain
Advantages for the physician:
Can be performed in patients for whom tissue biopsy is not possible
Allows real-time information through serial sampling
Molecular analyses of isolated cells provide insight into the disease and treatment response
Who Is Eligible?
CTC liquid biopsy is suitable for all cancer patients who are able to provide blood samples.
It is not performed in patients with hematological cancers.
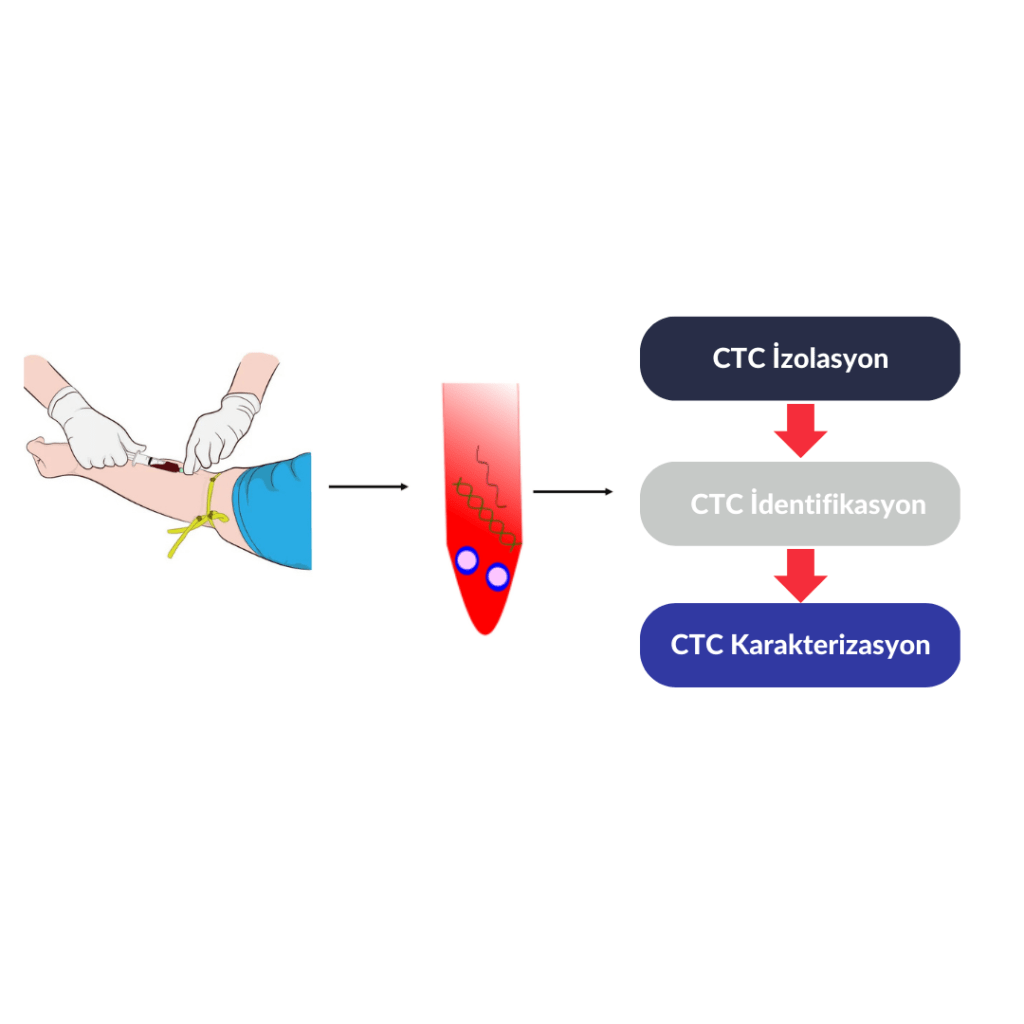
What Are the Steps?

Blood collection from the patient

Separation and identification of cancer cells from blood cells

Performing advanced molecular analyses on CTCs if deemed necessary by your physician

Preparation of the pathology report containing the results and delivery to your physician
The analysis results are delivered to your physician within two weeks.
This test is not a diagnostic test. The detection of CTCs through this test does not confirm a cancer diagnosis. However, in such a case, your physician may choose to monitor you more closely or refer you to additional diagnostic methods.
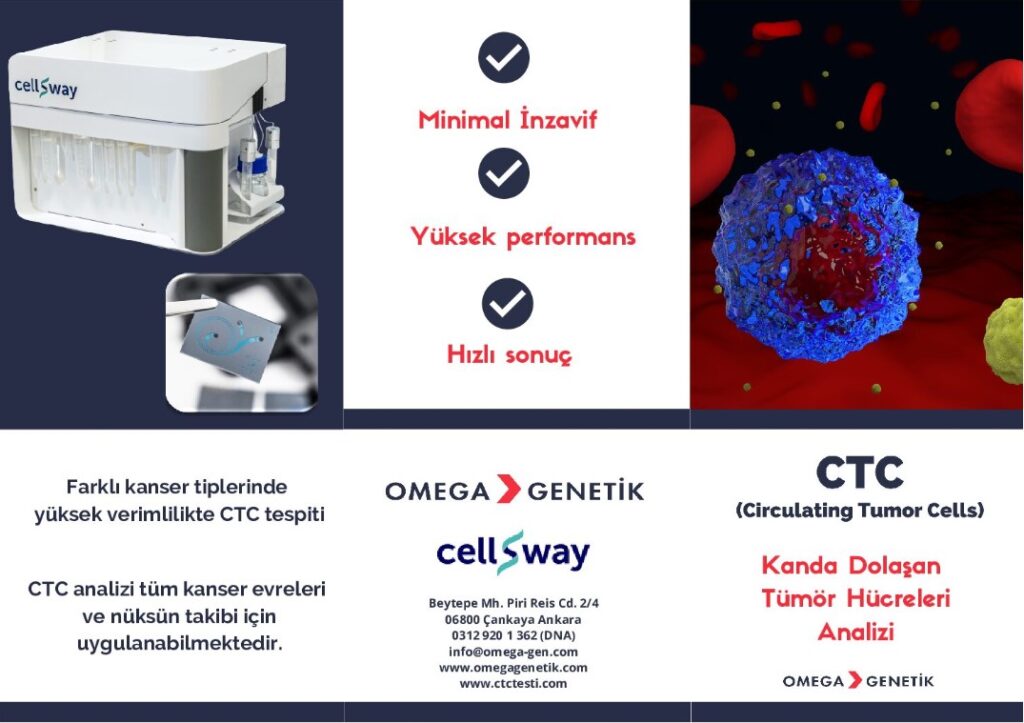
Cellsway CTC Analysis Device
The CTC analysis device, microfluidic chip, and test panels are for Research Use Only (RUO) and cannot be used for guiding cancer diagnosis or treatment.
The CTC analysis device developed by Cellsway enables the capture and analysis of tumor cells from the blood of patients diagnosed with cancer, using its unique microfluidic technology.

Cellsway CTC Analysis Device

Microfluidic Chip
Analytical Performance in Different Cancer Types
Analytical performance studies conducted in various cancer types—including lung, breast, prostate, colorectal, and ovarian cancers—have shown that CTCs can be captured with high efficiency. Clinical studies reported CTC positivity in 90% of prostate cancer patients and 60% of breast cancer patients. CTC analysis can be applied across all cancer stages and for monitoring recurrence.
Cellsway CTC Test Panels
The CTC analysis device, microfluidic chip, and test panels are for Research Use Only (RUO) and cannot be used for guiding cancer diagnosis or treatment.
CTC Identification Test Panel
The CTC Identification Test Panel enables the detection of epithelial-origin CTCs (CD45-, cytokeratin+, DAPI+) using immunofluorescent analysis.
The kit includes all necessary solutions for staining cytokeratin, CD45, and nuclei.
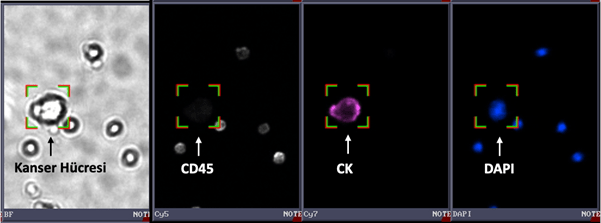
Breast cancer cell stained with the CTC Identification Test Panel
ER/PR/HER2 CTC Test Panel
The ER/PR/HER2 CTC Test Panel enables the determination of hormone receptor status—Estrogen Receptor (ER), Progesterone Receptor (PR)—and Human Epidermal Growth Factor Receptor 2 (HER2) in CTCs using immunofluorescent analysis. The status of these markers plays a particularly important role in breast cancer for understanding disease progression and guiding treatment decisions.
The kit includes all solutions necessary for ER, PR, and HER2 staining and is designed to be used in conjunction with the CTC Identification Kit.
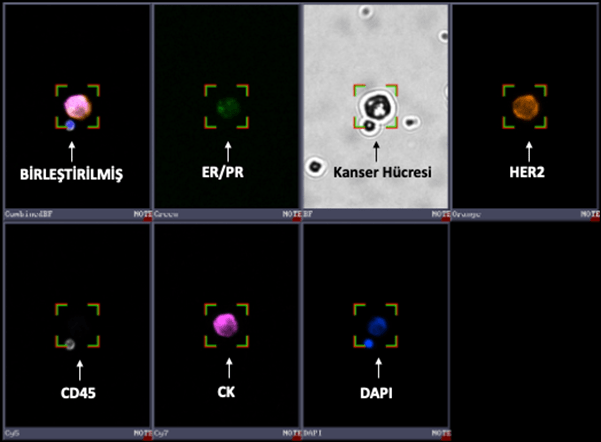
Breast cancer cell stained with the ER/PR/HER2 CTC Test Panel
PD-L1 CTC Test Panel
The PD-L1 CTC Test Panel enables the detection of the PD-L1 biomarker in CTCs using immunofluorescent analysis. It is known that patients with PD-L1 positivity respond better to immune checkpoint inhibitor therapies. The status of the PD-L1 marker plays a particularly important role in lung cancer for understanding disease progression and guiding treatment decisions.
The kit includes all solutions necessary for PD-L1 staining and is designed to be used in conjunction with the CTC Identification Kit.
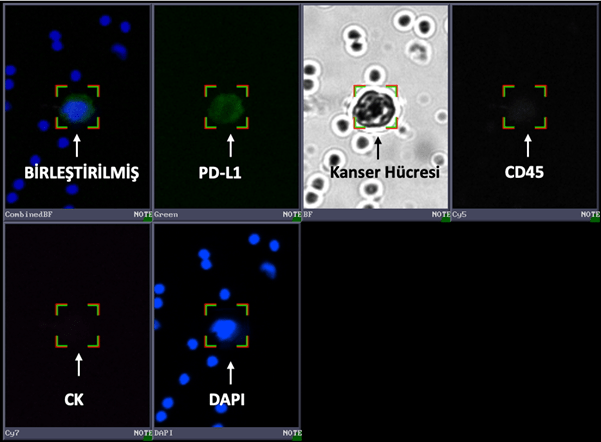
Breast cancer cell stained with the PD-L1 CTC Test Panel
CTC test panels are for Research Use Only (RUO) and cannot be used for guiding cancer diagnosis or treatment.